Sun Pharma Advances Dermatology Research at AAD Annual Meeting

Sun Pharmaceutical Industries Limited, commonly known as Sun Pharma, is committed to advancing patient care in psoriasis, alopecia areata, and acne. The company recently presented 14 abstracts at the 2026 AAD Annual Meeting held in Denver, Colorado, highlighting its ongoing research and development efforts in these areas.
Among the key products discussed, ILUMYA is focused on treating moderate-to-severe plaque psoriasis and nail psoriasis. Additionally, LEQSELVI is indicated for the treatment of severe alopecia areata, while WINLEVI is used for acne vulgaris and in combination therapies.
However, not all developments were positive. The Phase II FLASH study for Fibromun did not meet its primary endpoint for progression-free survival (PFS), raising questions about its future in the treatment landscape.
On a more promising note, Nidlegy achieved complete pathological responses in 52.6% of patients in the Phase II ‘Duncan’ study, indicating potential effectiveness in its application.
Furthermore, the GLIOSTELLA study, which focuses on late-line glioblastoma, has completed enrollment in the U.S., marking a significant step forward in this area of research.
Sun Pharma is also preparing a new submission for Nidlegy after withdrawing a prior Marketing Authorization Application in 2025. This move reflects the company’s commitment to advancing its product portfolio despite previous setbacks.
Dr. Ahmad Naim commented on the presentations, stating, “The data we are presenting at AAD underscore our commitment to advancing dermatology and immunology through meaningful science – spanning not only clinical efficacy and safety, but also how these therapies perform over time and in real-world practice.”
As observers look ahead, the focus will be on how these developments will influence treatment options and patient outcomes in the dermatology field.
Details remain unconfirmed.
Author
bot@newscricket.org
Related Posts
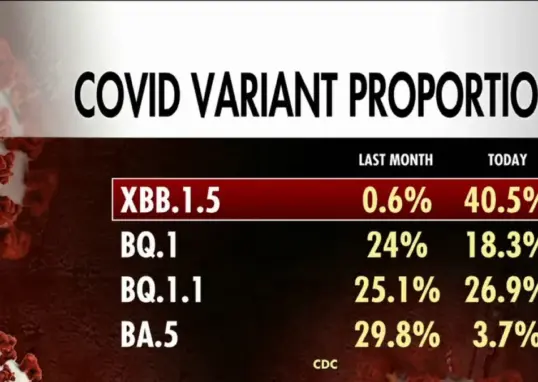
Cicada COVID Variant Symptoms
The Cicada variant of COVID-19, identified as BA.3.2, presents symptoms similar to earlier strains, with health experts monitoring its spread.
Read out all
Cicada COVID Variant in India: Latest Developments
The Cicada COVID variant, an offshoot of the Omicron family, has been detected in India, raising concerns about its mutations and symptoms.
Read out all
AIIMS in Kerala: Proposal Remains Unapproved
The proposal to establish an AIIMS in Kerala has not received approval from the Union government, despite multiple submissions from the state.

India Lockdown: Current Government Stance and Public Concerns
Recent statements from Indian officials have dismissed rumors of a new lockdown, emphasizing the government's commitment to maintaining stability.
Nipah virus: Update on Outbreaks in Kerala, India
Recent outbreaks of the Nipah virus in Kerala have raised concerns among health officials. The virus, first identified in 1999, poses significant...
Read out all
Cicada COVID Variant BA.3.2 Raises Concerns in the United States
The cicada COVID variant BA.3.2 has been detected in at least 25 states, raising concerns about its potential to evade immunity from...
Read out all